The Clinical Hematopathology Laboratory offers several different tests for the diagnosis and characterization of hematopoietic malignancy. The principles underlying these tests are described below, together with a list of publications demonstrating their application and clinical utility.
Tests
Immunophenotyping by Flow Cytometry
Flow cytometry involves staining live cells with fluorescently labeled antibodies that bind to proteins expressed on the cell surface. Different types of lymphocytes express different proteins (for example, T-cells express the protein CD3, and B cells express the protein CD21). The cells are analyzed on a flow cytometer, which tells us how many cells of each type are present. This information allows us to determine the lineage of the cells present, and whether they are homogeneous (more consistent with neoplasia) or heterogeneous (more consistent with a reactive process).
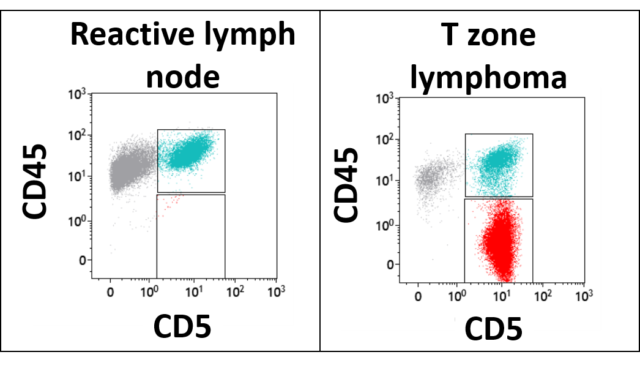
The image below demonstrates how T zone lymphoma is diagnosed by flow cytometry. In these graphs, each dot represents a cell. The cells are plotted based on their level of expression of the T cell antigen CD5 on the X axis, and the level of their expression of the pan-leukocyte antigen CD45 on the Y axis. The graph on the left shows that a reactive lymph node is a mixture of T cells (in green) and non-T cells (in gray). All the cells in this lymph node express CD45. The graph on the right is from a dog with T zone lymphoma. In this lymph node, most of the cells express CD5, but a significant subpopulation of these cells have lost expression of CD45. This observation is considered diagnostic for T zone lymphoma.
Polymerase chain reaction for Antigen Receptor Rearrangement (PARR)
The PARR assay is a PCR assay in which the DNA of immunoglobulin (antibody) and T cell receptor genes is amplified. The results tell us if the majority of cells in the sample are derived from a single clone (most consistent with neoplasia), or from multiple clones (most consistent with a reactive process).
We estimate we detect 93% of confirmed canine lymphomas and 91% of confirmed feline lymphoma cases with the PARR assay. Therefore, a negative result does not rule out a diagnosis of lymphoma in either species.
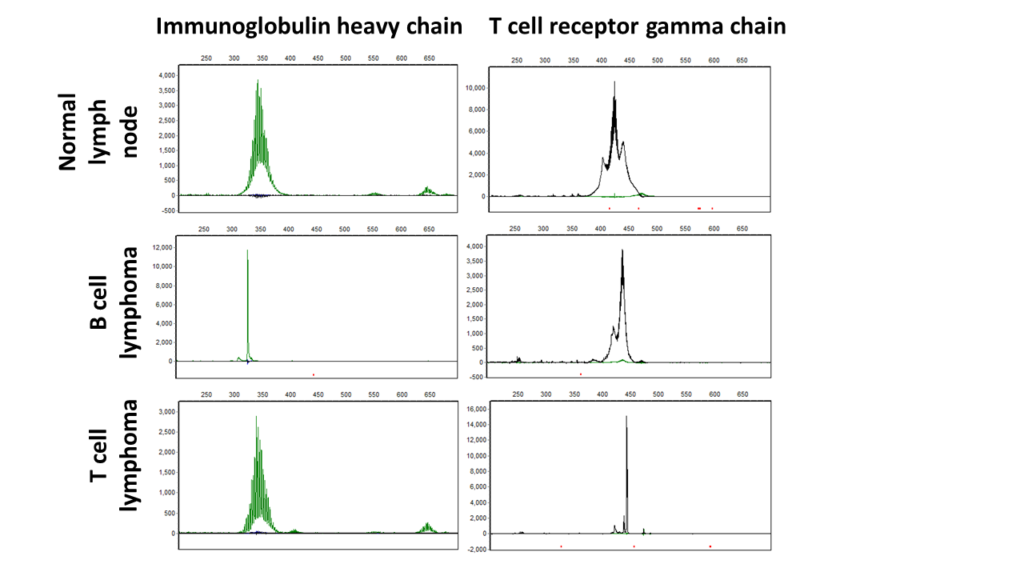
The figure below shows PARR assay results for a normal lymph node (top row), B-cell lymphoma (middle row) and T-cell lymphoma (bottom row). The B-cell lymphoma and T-cell lymphoma have single sized PCR products in the immunoglobulin gene reaction (left column) and T-cell receptor reaction (right column), respectively.
Ki67
Ki67 is a protein that is expressed in the nucleus of a cell during proliferation but is absent when cells are quiescent. It is a commonly used marker of proliferation in immunohistochemistry. In some cancers, high Ki67 expression predicts a more aggressive disease.
We have found that Ki67 can help to distinguish cases of B-cell chronic lymphocytic leukemia with a short survival from those with a more indolent clinical course. Testing for Ki67 is not intended to guide therapy, but to provide some additional prognostic information about this disease, which has a very heterogeneous clinical course. Ki67 is detected using a flow cytometry assay. We have not established the value of Ki67 detection in other forms of lymphoid neoplasia.
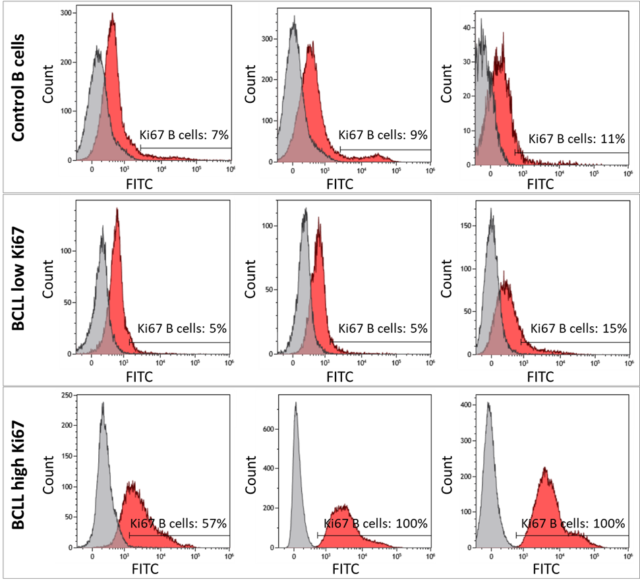
The figure below shows flow cytometry of Ki67 expression in B-cells from control dogs and from dogs with B-cell chronic lymphocytic leukemia (BCLL). Representative plots are shown from 3 control dogs without lymphoproliferative disease (2 lymph node samples [top left, top center] and 1 blood sample [top right]), 3 BCLL cases with low Ki67 expression (middle), and 3 BCLL cases with high Ki67 expression (bottom). Samples were stained with anti-Ki67-FITC (red histogram) or isotype-FITC control (grey histogram) and CD21+ B-cells were gated for flow cytometry analysis. The isotype control was strictly used to determine the percent positive for Ki67 expression. Control B-cells and low Ki67 BCLL cases had <20% Ki67-expressing B-cells. High Ki67 BCLL cases had >40% Ki67-expressing B-cells.
Cell Block
Cell block techniques can be used to fix fluid samples (i.e. blood, effusions, or aspirates) into a paraffin block that can be used for immunohistochemical staining. This technique can increase diagnostic accuracy by applying additional antibodies that are not readily available for flow cytometry and allow us to investigate expression of intracellular antigens. Immunohistochemistry typically provides clearer and more consistent results than immunocytochemistry.
Utility of cell block
Occasionally, we will not be able to characterize a hematopoietic neoplasm based on the readily available antibodies used for flow cytometry. Cell blocks are useful for investigating the expression of additional antigens in lineage ambiguous hematopoietic neoplasms, particularly those found in the cytoplasm or nucleus of cells. For example, we currently do not have an antibody that can recognize surface antigens that are specific for plasma cells. Therefore, we can use cell block techniques to determine if the cells express intracellular IRF4/MUM1, an antigen associated with plasma cell differentiation. Cell blocks are not limited to plasma cell neoplasms and we can apply additional antibodies that are available for immunohistochemistry. Other antigens detected include, but aren’t limited to: CD204, CD18, IBA1, CD20, PAX5 and CD3.
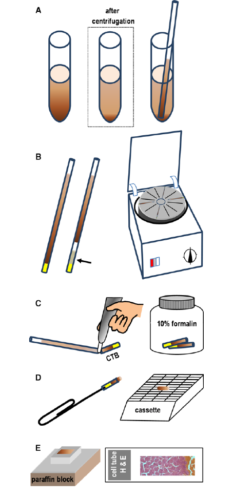
Schematic overview of cell block technique
- (A) A capillary tube is filled with liquid or a sediment (in low cellular samples).
- (B) Capillary tubes are sealed with clay; in the absence of significant blood contamination, a high-density medium (eg, percoll) may be aspirated to facilitate separation of the cell pellet from the clay (arrow).
- (C) After centrifugation, the capillary tube is broken at the liquid–solid interface using a glass-writing diamond pen.
- (D) After fixation in formalin for at least 24 hours, the CTB is pushed out of the glass capillary using a paper clip and placed in a cassette for routine paraffin embedding.
- (E) Paraffin-embedded CTB sections are stained with H&E or other stains
Case study example
The blood from a 7-year-old English setter was analyzed by flow cytometry and expressed CD34 but no other lineage-specific antigens, leading to a diagnosis of acute leukemia. The lineage (myeloid vs lymphoid) was not clear from flow cytometry. The buffy coat was embedded in a paraffin block and stained with standard immunohistochemical procedures. The cells express the T-cell antigen CD3 (brown staining) but not B-cell antigens CD20 and PAX5, nor the plasma cell antigen MUM1. The expression of CD3 indicates T-cell lineage (acute T-cell leukemia). CD3 was not detected by flow cytometry because acute T-cell leukemias typically do not express surface CD3, but will express cytoplasmic CD3 which is detected by immunohistochemistry.

Publications
Our publications describe the assays used in the laboratory, how they are used to provide prognostic information, and the spectrum of lymphoproliferative disorders in dogs and cats.| Author | Publication Date | Article Title | Journal | Full Article |
|---|---|---|---|---|
| Harris, R.A. et al | 2022 | Using digital RNA counting to establish flow cytometry diagnostic criteria for subtypes of CD34+ canine acute leukaemia | Vet Comp Oncol | View Full Article |
| Rout, E.D. et al | 2022 | Clinical outcome and Ki67 evaluation in dogs with nodal small cell B-cell lymphoma diagnosed by flow cytometry | J Vet Intern Med | View Full Article |
| Frankhouse, K. et al | 2021 | An aggressive CD4-CD8- T-cell neoplasm in young English bulldogs | Vet Comp Oncol | View Full Article |
| Hughes, K.L. | 2021 | Diffuse small B-cell lymphoma: A high-grade malignancy | Vet Path | View Full Article |
| Rout, E.D. et al | 2021 | Clinical outcome and prognostic factors in dogs with B-cell chronic lymphocytic leukemia: A retrospective study | J Vet Intern Med | View Full Article |
| Rout, E.D. et al | 2020 | Polyclonal B-cell lymphocytosis in English bulldogs | J Vet Intern Med | View Full Article |
| Labadie, J.D. et al | 2020 | Genome-wide association analysis of canine T zone lymphoma identifies link to hypothyroidism and a shared association with mast-cell tumors | BMC Genomics | View Full Article |
| Harris, L.J. et al | 2020 | Clinical features of canine nodal T-cell lymphomas classified as CD8+ or CD4−CD8− by flow cytometry | Vet Comp Oncol | View Full Article |
| Labadie, J.D. et al | 2019 | Associations of environment, health history, T-zone lymphoma, and T-zone-like cells of undetermined significance: A case-control study of aged Golden Retrievers | J Vet Intern Med | View Full Article |
| Rout, E.D. et al | 2019 | Immunophenotypic characterization and clinical outcome in cats with lymphocytosis | J Vet Intern Med | View Full Article |
| Wolff-Ringwall, A. et al | 2019 | Prospective evaluation of flow cytometric characteristics, histopathologic diagnosis and clinical outcome in dogs with naïve B-cell lymphoma treated with a 19-week CHOP protocol | Vet Comp Oncol | View Full Article |
| Harris, L.J. et al | 2019 | Canine CD4+ T-cell lymphoma identified by flow cytometry exhibits a consistent histomorphology and gene expression profile | Vet Comp Oncol | View Full Article |
| Rout, E.D. et al | 2019 | Assessment of immunoglobulin heavy chain, immunoglobulin light chain, and T‐cell receptor clonality testing in the diagnosis of feline lymphoid neoplasia | Vet Clin Pathol | View Full Article |
| Rout, E.D. et al | 2018 | Preferential use of unmutated immunoglobulin heavy variable region genes in Boxer dogs with chronic lymphocytic leukemia | PLoS One | View Full Article |
| Wiles, V. et al | 2018 | Metastatic thymoma in the liver of a dog | J Vet Diag Invest | View Full Article |
| Harris, L.J. et al | 2017 | Clinicopathologic features of lingual canine T-zone lymphoma | Vet Comp Oncol | View Full Article |
| Rout, E.D. et al | 2017 | Progression of cutaneous plasmacytoma to plasma cell leukemia in a dog | Vet Clin Pathol | View Full Article |
| Rout, E.D. et al | 2017 | Lymphoid neoplasia correlations between morphology and flow cytometry | Vet Clin Small Anim | View Full Article |
| Hughes, K.L. et al | 2017 | Increased frequency of CD45 negative T cells (T zone cells) in older Golden retriever dogs | Vet Comp Oncol | View Full Article |
| Bromberek, J. et al | 2016 | Breed distribution and clinical characteristics of B-cell chronic lymphocytic leukemia in dogs | J Vet Intern Med | View Full Article |
| Avery, P.R. et al | 2014 | Flow cytometric characterization and clinical outcome of CD4+ T-cell lymphoma in dogs: 67 cases | J Vet Intern Med | View Full Article |
| Seelig, D.M. et al | 2014 | Canine T zone lymphoma: a unique immunohenotypic features, outcome and population characteristics | J Vet Intern Med | View Full Article |
| Rao, S. et al | 2011 | Class II major histocompatibility complex expression and cell size independently predict survival in canine B-cell lymphoma | J Vet Intern Med | View Full article |
| Williams, M. et al | 2008 | Canine lymphoproliferative disease characterized by lymphocytosis: immunophenotypic markers of prognosis | J Vet Intern Med | View Full Article |
| Lana, S. et al | 2006 | Diagnosis of mediastinal masses in dogs by flow cytometry | J Vet Intern Med | View Full Article |